Child cancer trials 2025, Trump administration impact, Brain cancer research halt, Sec Kennedy actions, MAHA crew decisions

The trump administration is killing off clinical trials for kids with brain cancer. This is what @SecKennedy and the MAHA crew is actually doing. No one voted for this. No one. pic.twitter.com/TgSoWnixkB
— Tommy Vietor (@TVietor08) August 29, 2025
- YOU MAY ALSO LIKE TO WATCH THIS TRENDING STORY ON YOUTUBE. Waverly Hills Hospital's Horror Story: The Most Haunted Room 502
The Impact of the Trump Administration on Pediatric Brain Cancer Clinical Trials
The landscape of pediatric medicine has been significantly influenced by the policies and decisions made during the Trump administration, particularly in regard to clinical trials for children suffering from brain cancer. A recent tweet from Tommy Vietor has resurfaced the conversation around this vital issue, highlighting concerns over the administration’s stance and actions that some believe have detrimental effects on the future of clinical research and potential treatments for young patients.
Understanding Pediatric Brain Cancer
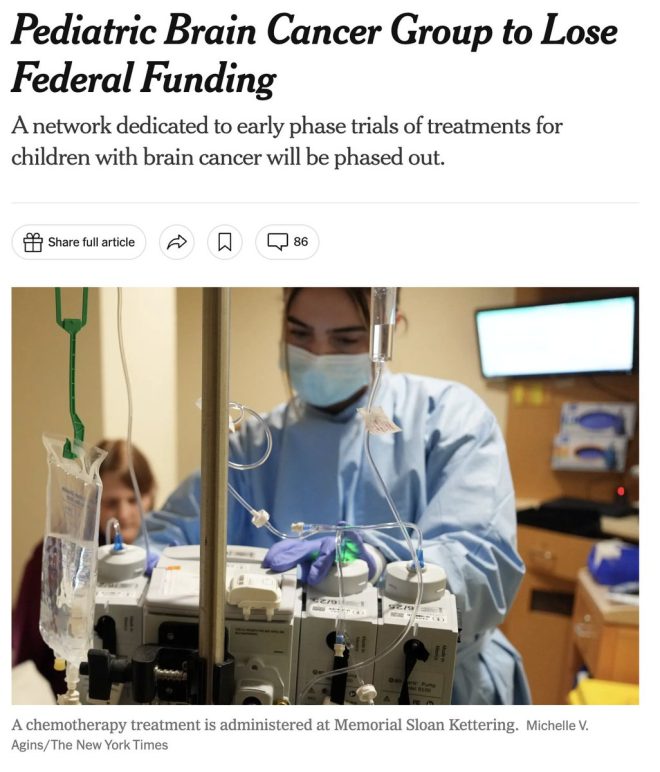
Pediatric brain cancer represents one of the most challenging areas in oncology. Brain tumors in children can vary widely in type, location, and aggressiveness, making treatment particularly complex. Standard treatments often include surgery, radiation, and chemotherapy, but the efficacy varies based on the tumor type and the child’s unique circumstances.
Clinical trials play a crucial role in developing new and innovative treatments, offering hope to families who are facing dire circumstances. These trials not only test the efficacy of new therapies but also provide critical data on how these treatments affect children differently compared to adults.
The Role of Clinical Trials in Advancing Treatment
Clinical trials are essential for advancing medical knowledge and improving treatment options. They allow researchers to evaluate new drugs, therapies, and interventions before they receive approval for general use. For pediatric brain cancer, which often lacks sufficient funding compared to adult cancers, clinical trials are particularly vital. They can lead to breakthroughs that improve survival rates and quality of life for young patients.
The Trump Administration’s Policies and Their Consequences
The tweet by Vietor raises alarm over recent policies enacted by the Trump administration that are perceived to impede the progress of clinical trials for children with brain cancer. Critics argue that the administration’s actions have led to a decrease in funding and support for pediatric research, resulting in fewer clinical trials being conducted. This, in turn, limits the treatment options available to children suffering from these aggressive forms of cancer.
Many believe that the decision to cut funding for specific research initiatives is not only a political misstep but also a moral one, as it directly affects the health and well-being of vulnerable children. The notion that no one voted for these policies underscores a growing frustration among healthcare advocates, families, and researchers who feel that the voices of those affected by pediatric cancer are not being heard.
The Importance of Advocacy and Awareness
The situation highlights the need for advocacy and awareness surrounding pediatric brain cancer and the importance of research funding. Advocacy groups and individuals play a crucial role in pushing for policy changes that prioritize the needs of children with cancer. By raising awareness, they can mobilize public support and influence decision-makers to allocate necessary resources for research and clinical trials.
Social media platforms, like Twitter, have become powerful tools for spreading awareness and galvanizing support. Vietor’s tweet serves as a rallying cry for those in the fight against pediatric brain cancer, reminding the public of the urgent need for continued research and innovation.
Moving Forward: Solutions and Strategies
To address the challenges faced by pediatric clinical trials, several strategies can be implemented:
- Increased Funding for Pediatric Research: Governments and private organizations need to prioritize funding for pediatric cancer research. This includes supporting both large-scale initiatives and smaller, innovative trials that target specific types of brain tumors.
- Collaboration Between Organizations: Collaboration between different stakeholders—such as hospitals, research institutions, and advocacy groups—can enhance the effectiveness of clinical trials. By pooling resources and expertise, these collaborations can accelerate the development of new therapies.
- Public Awareness Campaigns: Raising awareness about the importance of clinical trials for pediatric brain cancer can mobilize public support and encourage donations to research initiatives. Awareness campaigns can also educate families about the availability of clinical trials and how they can participate.
- Policy Advocacy: Engaging in advocacy at local, state, and national levels can help influence policy changes that support pediatric cancer research. This includes lobbying for policies that increase funding and streamline the process for conducting clinical trials.
- Patient-Centric Approaches: Ensuring that clinical trials are designed with the needs of pediatric patients in mind can improve enrollment and retention rates. This includes considering the unique challenges faced by children and their families when participating in trials.
Conclusion
The ongoing conversation surrounding clinical trials for pediatric brain cancer is a critical one. It underscores the intersection of healthcare policy, research funding, and the well-being of children battling this devastating illness. As highlighted by Tommy Vietor’s tweet, the implications of political decisions can have far-reaching effects, particularly on vulnerable populations like children with cancer.
Advocacy, awareness, and strategic action are essential to ensure that pediatric brain cancer research remains a priority. By working together—researchers, policymakers, and the public—there is hope for breakthroughs that can lead to improved outcomes for children facing this challenging diagnosis. The fight against pediatric brain cancer is not just a medical challenge; it is a societal obligation that demands collective action and unwavering commitment.

Trump Admin Ends Kids’ Brain Cancer Trials: Who Voted?
” /> 
The Trump administration is killing off clinical trials for kids with brain cancer. This is what @SecKennedy and the MAHA crew is actually doing. No one voted for this. No one. pic.twitter.com/TgSoWnixkB
— Tommy Vietor (@TVietor08) August 29, 2025
The Trump Administration is Killing Off Clinical Trials for Kids with Brain Cancer
The landscape of cancer treatment has always been complex, but recent actions have stirred a significant debate regarding clinical trials for children battling brain cancer. The statement by Tommy Vietor, highlighting that *The Trump administration is killing off clinical trials for kids with brain cancer*, raises critical concerns that demand our attention. These trials are often the last hope for families facing this devastating disease, and any disruption can have dire consequences.
This is What @SecKennedy and the MAHA Crew are Actually Doing
In the tweet, Vietor specifically points fingers at @SecKennedy and the MAHA crew, suggesting that their actions are detrimental to these vulnerable patients. It’s essential to understand what MAHA stands for; it’s the *Medical and Health Affairs* group that has a role in healthcare decisions. The claim here is serious: by hindering clinical trials, they might be jeopardizing the future of innovative treatments that could save lives.
When we talk about clinical trials, we’re discussing rigorous tests designed to evaluate new treatments. These trials are crucial for understanding the effectiveness and safety of therapies, especially in pediatric cases where options can be limited. The implications of halting these trials are profound, as they can stifle breakthroughs in treatment that could alleviate suffering for countless children and their families.
No One Voted for This. No One.
Vietor’s strong statement that “No one voted for this” resonates deeply with many families who are directly affected by brain cancer. It implies a disconnect between the decision-makers and the real-world implications of their policies. Parents and families are left feeling helpless, navigating a system that seems to have forgotten their needs.
The phrase serves as a rallying cry for accountability. When it comes to the health and well-being of children, it’s crucial that those in power listen to the voices of affected families. The decisions made by leaders should reflect the needs of the constituents they serve, particularly when those constituents are the most vulnerable among us, like kids fighting cancer.
The Importance of Clinical Trials in Pediatric Oncology
Clinical trials for pediatric patients are essential for several reasons. First, children’s bodies react differently to treatments than adults. What works for an adult may not work for a child, which is why tailored clinical trials are critical. They offer a pathway to discover new therapies that are more effective and have fewer side effects.
Moreover, these trials often provide access to cutting-edge treatments that aren’t yet available through traditional channels. For families facing brain cancer, this could mean the difference between life and death. The emotional toll on families waiting for new hope can be immense. With clinical trials, there’s a chance for not just hope, but tangible progress in treating this devastating disease.
The Fallout of Canceling Clinical Trials
If the claims about the Trump administration’s actions are accurate, we could witness severe fallout. Canceling or hindering clinical trials could lead to a stagnation in research and a lack of new therapies. For children with brain cancer, this could mean fewer options and increased suffering.
Families often rely on the promise of future treatments to cope with the diagnosis of brain cancer. When that promise is taken away, it can lead to despair and frustration. Advocacy groups, researchers, and healthcare professionals are already sounding the alarm, urging for the continuation and expansion of these trials instead of their cancellation.
The Role of Advocacy in Healthcare Policy
Advocacy plays a pivotal role in shaping healthcare policy, especially regarding vulnerable populations like children with cancer. Organizations dedicated to pediatric oncology work tirelessly to ensure that clinical trials remain a viable option. This is where community support and awareness come into play.
By mobilizing public opinion and raising awareness, advocates can influence policymakers to reconsider decisions that adversely affect clinical trials. Social media has become a powerful tool for advocacy, allowing families and supporters to share their stories and push for change. The tweet from Vietor serves as an example of how digital platforms can amplify voices and generate important conversations around healthcare policy.
What Can You Do? Get Involved!
If you’re concerned about the potential cancellation of clinical trials for kids with brain cancer, there are several steps you can take to get involved. First, educate yourself about the current state of pediatric oncology and the importance of clinical trials. Knowledge is power, and understanding the issue can help you advocate more effectively.
Consider supporting organizations that focus on pediatric cancer research. Donations can help fund trials and ensure that research continues without interruption. You can also participate in awareness campaigns, share information on social media, and contact your local representatives to voice your concerns. Every action counts, and collective efforts can lead to significant changes.
Taking a stand for children’s health is imperative, and it starts with being informed and involved. The future of clinical trials for pediatric brain cancer patients may depend on it.
Final Thoughts
The conversation surrounding clinical trials for kids with brain cancer is more than just a policy debate; it’s about real lives at stake. The message from Tommy Vietor highlights the urgency of the situation and the need for accountability from those in power. As advocates for change, we must ensure that our most vulnerable populations receive the care and attention they deserve.
By staying informed, supporting research, and advocating for continued clinical trials, we can contribute to a future where effective treatments for brain cancer are accessible to all children.
brain cancer research funding, pediatric oncology trials 2025, Trump administration health policies, childhood cancer advocacy, Sec Kennedy healthcare decisions, MAHA group actions, clinical trial access for children, pediatric brain tumor studies, healthcare legislation impact, cancer treatment approval process, Trump administration healthcare changes, patient rights in clinical trials, government funding for cancer research, childhood cancer survival rates, advocacy for pediatric patients, brain cancer treatment innovations, healthcare policy controversies, clinical research ethics, legislative impact on healthcare, children’s health rights
